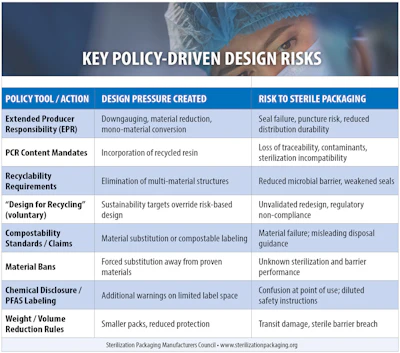
Sustainable packaging policy is reshaping packaging decisions across the United States, and despite some exemptions in certain cases, healthcare packaging is increasingly affected. Extended Producer Responsibility (EPR) programs, recycled content mandates, recyclability requirements, compostability standards, and chemical disclosure laws are designed to reduce waste and advance circularity. Yet for sterile medical device packaging, these policies introduce design pressures that can directly conflict with regulatory and performance requirements ensuring safety remains of paramount importance.
The Sterilization Packaging Manufacturers Council (SPMC) has examined these conflicts in depth and is working on a series of white papers addressing this topic, starting with Sustainability of Medical Device Packaging. The findings are clear: sterile packaging is a safety-critical system governed by FDA regulations and international standards, and sustainability policies that fail to account for these requirements can introduce unacceptable risk.
Medical device packaging must meet stringent obligations under FDA Quality System Regulations and consensus standards such as ISO 11607 for package integrity and ISO 10993 for biocompatibility. Policies requiring post-consumer recycled (PCR) content or universal recyclability are incompatible with these standards today. Mechanically recycled materials lack traceability, introduce contamination risk, and exhibit inconsistent performance that cannot be reliably validated for sterilization, seal integrity, or long-term barrier protection. In sterile packaging, even minor material variability can lead to seal failures, microbial ingress, and Class I recalls.
EPR laws illustrate how these pressures manifest in practice. Seven states—California, Colorado, Maine, Maryland, Minnesota, Oregon, and Washington—have enacted packaging EPR frameworks. While several states recognize the need to exclude sterile medical packaging, others leave ambiguity, particularly for secondary and tertiary packaging. Even where exemptions exist, EPR fee structures, recyclability metrics, and corporate sustainability commitments often drive voluntary redesigns that reduce material thickness, favor mono-material structures, or encourage recycled content—sometimes without sufficient risk evaluation.
To address this challenge, SPMC advocates for a risk-based sustainability assessment model grounded in regulatory compliance and patient safety. This model begins by asking a fundamental question: does a proposed sustainability change preserve sterility assurance and regulatory compliance across the entire lifecycle of the device? If not, it should not proceed.
Under this framework, sustainability initiatives must be evaluated against four core criteria: sterilization compatibility, microbial barrier performance, physical protection during distribution, and biocompatibility. Changes to materials, structures, or labeling should trigger formal validation and risk management processes consistent with FDA and ISO expectations.
Importantly, SPMC also recognizes the need for a strong advocate and educator on sterile medical packaging. Our industry needs to help legislators, regulators and key stakeholders understand and protect the value of sterile packaging systems, recognize where these policies can create failures and advocate for certain technologies and solutions to offer a safer pathway to circularity in the future.
Sustainable progress in healthcare packaging is both necessary and achievable—but only if policy, design, and sustainability goals are aligned with the realities of sterile packaging performance. As SPMC’s work underscores, patient safety must remain the non-negotiable foundation upon which all sustainability efforts are built.