This content was written and submitted by the supplier. It has only been modified to comply with this publication’s space and style.
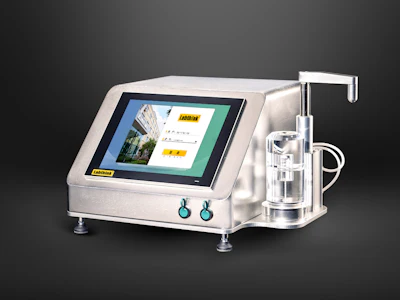
Labthink is proud to announce the launch of the C690M Nondestructive Package Leakage Detector, an advanced testing solution designed to ensure the integrity of pharmaceutical packaging. Based on the vacuum decay and pressure decay testing methods, the C690M is engineered and manufactured in accordance with ASTM F2338 and other internationally recognized standards.
The Labthink C690M is specifically developed for trace leak detection in pharmaceutical packaging, including vials, ampoule bottles, cartridge bottles, infusion bottles, and prefilled syringes. By providing accurate, repeatable, and nondestructive testing, the system helps pharmaceutical manufacturers maintain product safety, regulatory compliance, and packaging quality.
Advanced Multi-Stage Vacuum Technology
The C690M features a dual-method testing system using both vacuum decay and pressure decay methods, enabling flexible testing across a wide range of package types.
Key capabilities include:
- Automatic clamping system that securely fastens the test chamber for reliable testing
- Adjustable target vacuum levels allowing operators to meet specific detection requirements for different samples
- Custom transparent test chamber that enables clear observation of the sample during testing
- Optional built-in European oil-free vacuum pump, offering maintenance-free and contamination-free operation
- Advanced pressure detection technology using world-renowned components for stable data unaffected by ambient environmental conditions
- Automatic microflow control technology that simulates different leakage hole sizes without manual adjustment
Intelligent System Control
The C690M is equipped with an intuitive control platform designed for efficiency and ease of use.
Features include:
- 12.1-inch embedded touch tablet computer running Windows OS
- Real-time pressure curve display for clear monitoring of the testing process
- Automatic calculation of leakage rates and statistical analysis of results
- Compatibility with universal printers for convenient result output
- USB and network ports enabling external access, data transmission, and remote system upgrades
Compliance with Pharmaceutical Industry Standards
Designed with regulatory compliance in mind, the C690M meets the strict requirements of the pharmaceutical industry.
Compliance features include:
- Verification through compensation and calibration methods
- Full support for GMP data traceability requirements
- Multi-level user permission management with customizable access rights